Step 1 — Discover & Configure
(Week 1)
Map workflows, roles, and controls tailored to your operating model.
Includes:
- Requirements assessment
- Process mapping
- Initial configuration
Complere connects your quality records, actions, people, and evidence into one visible operating system—so you don't just pass audits, you understand and control quality in real time.
Records Actions People Evidence Real-time visibility Decision-ready control
Built for quality leaders who need control, visibility, and inspection confidence across regulated operations.
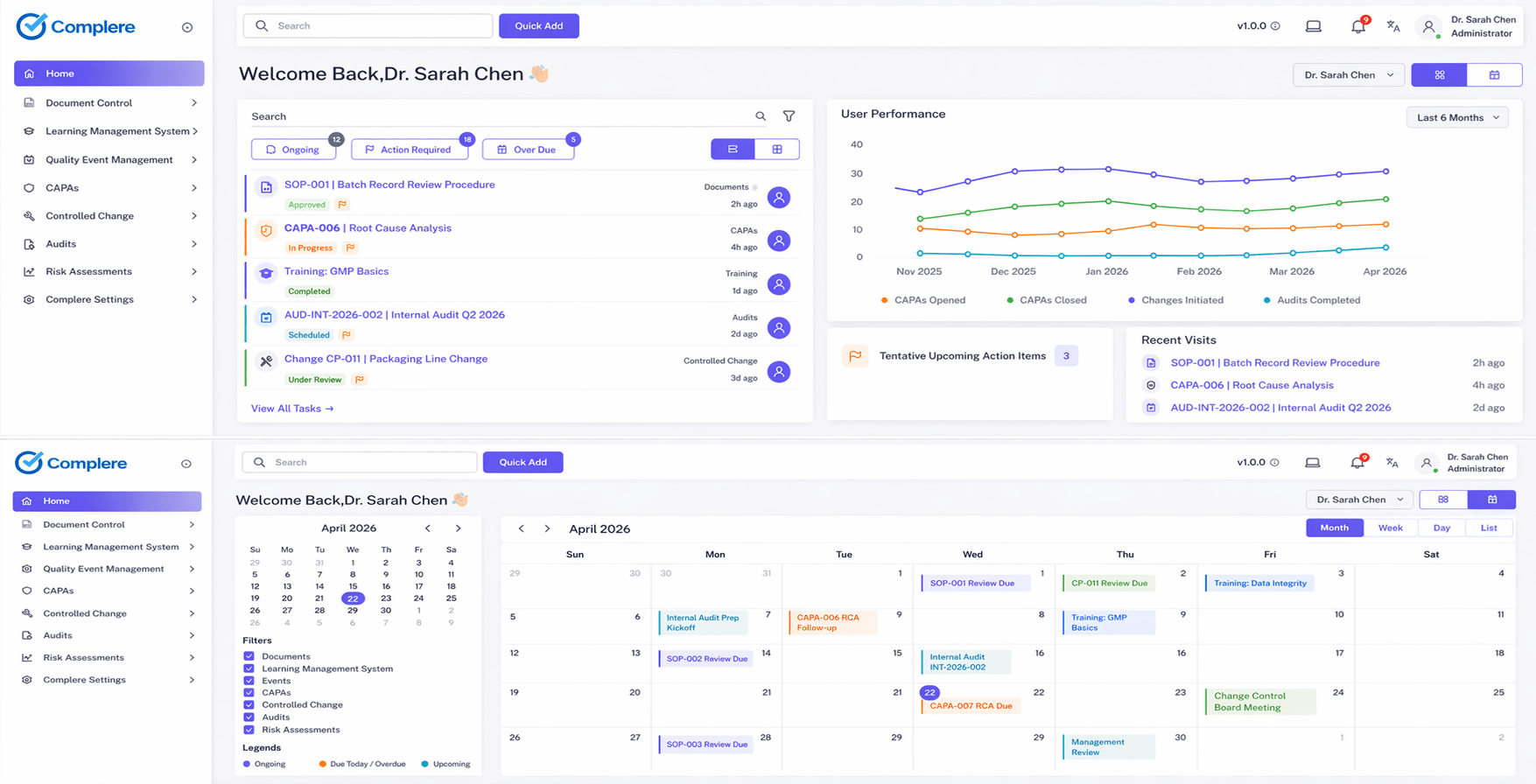
Designed for teams that cannot afford fragmented quality execution
Guided rollout with real workflows—reach usable, auditable outcomes in record time.
(Week 1)
Map workflows, roles, and controls tailored to your operating model.
Includes:
(Weeks 2–4)
Run live workflows to validate traceability and readiness for compliance.
Includes:
(Week 5)
Confirm readiness, finalize production rollout, and provide hypercare.
Includes:
See traceability, usability, and inspection readiness using your processes.
Typical pilot: 4–6 weeks depending on scope and data readiness.
Apply the right level of validation rigor based on risk—without slowing adoption.
Documented IQ/OQ/PQ evidence, traceable requirements, and release discipline aligned to how QA and IT qualify systems.
Focus testing on what matters for patient safety, product quality, and data integrity—while keeping records inspection-defensible.
Controlled access and approvals by role, site, and function.
Time-stamped, immutable history for governed changes.
Before-and-after visibility for documents and critical records.
Restore with audit context instead of silent data loss.
Complere is shaped by people with direct experience in pharmaceutical quality operations, medical device compliance, and GxP validation — not generic enterprise software marketing.
Across pharmaceutical manufacturing, biotech, medical devices, and life sciences quality operations — including inspection support, deviations, and validation programmes.
We engage with the same professional communities and guidance frameworks — GAMP 5, data integrity, and computerized systems — that quality and IT leaders use to qualify vendors.
VMP, URS, risk assessment, and IQ/OQ/PQ templates ship with deployment — structured for regulated rollout, not sold as a separate line item.
Teams report shorter retrieval time for SOPs, training records, and CAPA chains when asked to show evidence during audits or customer QA reviews.
Structured IQ/OQ/PQ alignment with documented roles, plus support for release impact when the platform or configuration changes.
Review the validation approach and Annex 11 validation playbook before your formal assessment.
Anonymized examples illustrate typical scope, timeline, and measurable benefits — your account team can share sector-relevant detail during qualification.
Scope: document control, training, CAPA, change control.
Timeline: pilot in one site, multi-site rollout in phases.
Outcome: consolidated audit trail for procedures and training evidence; fewer overdue training findings in internal audits.
Scope: deviations, CAPA, supplier quality linkage.
Timeline: core workflows live before pivotal trial supply milestones.
Outcome: traceable investigation records and effectiveness checks for regulatory file readiness.
Scope: change control, document control, training.
Timeline: aligned with design transfer window.
Outcome: linked change records, updated procedures, and training completion before product release.
Connected quality modules spanning the full quality lifecycle—each with the core features that keep your system inspection-ready and practical for regulated teams.
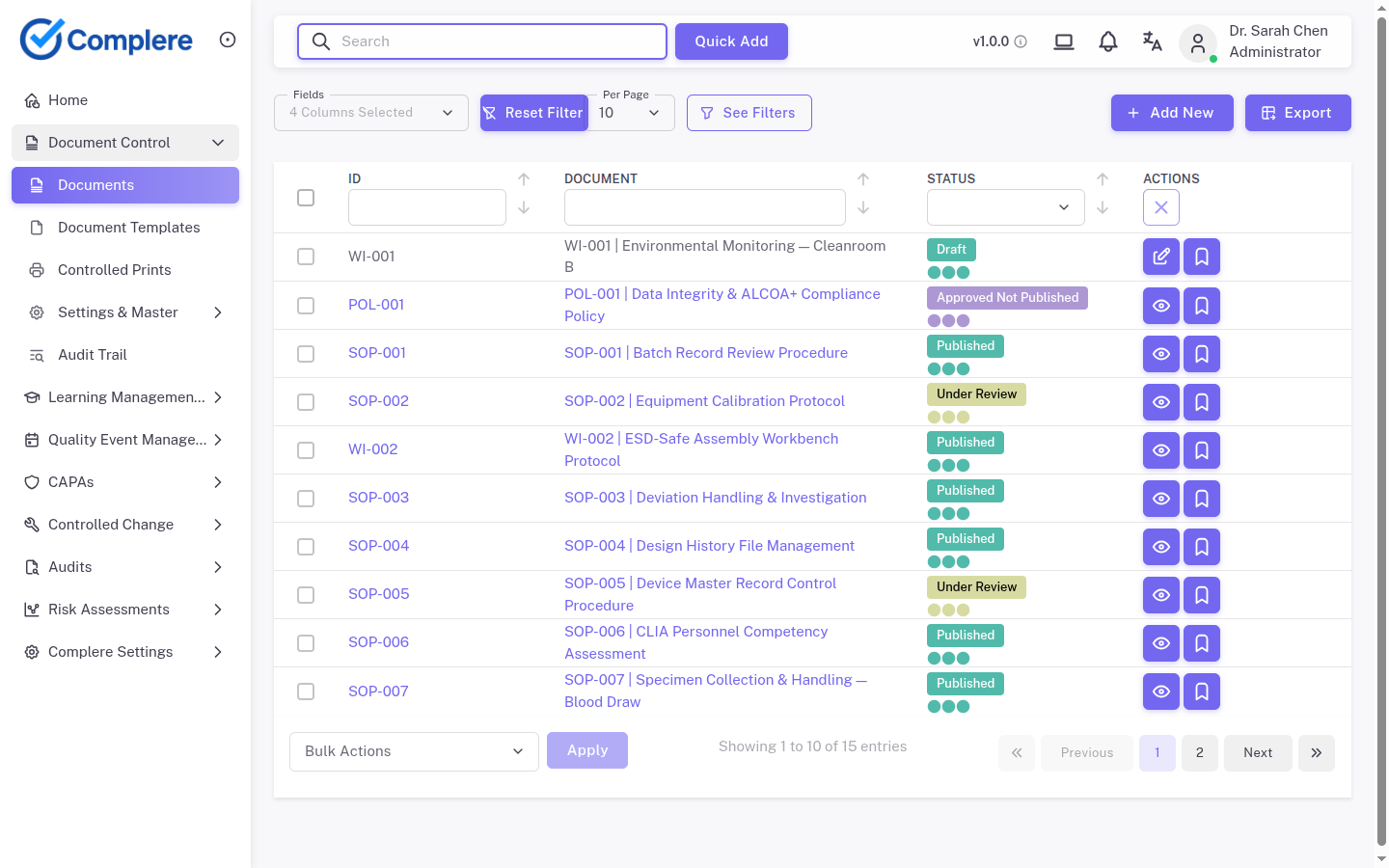
Control SOPs, forms, templates, and approvals without relying on shared drives.
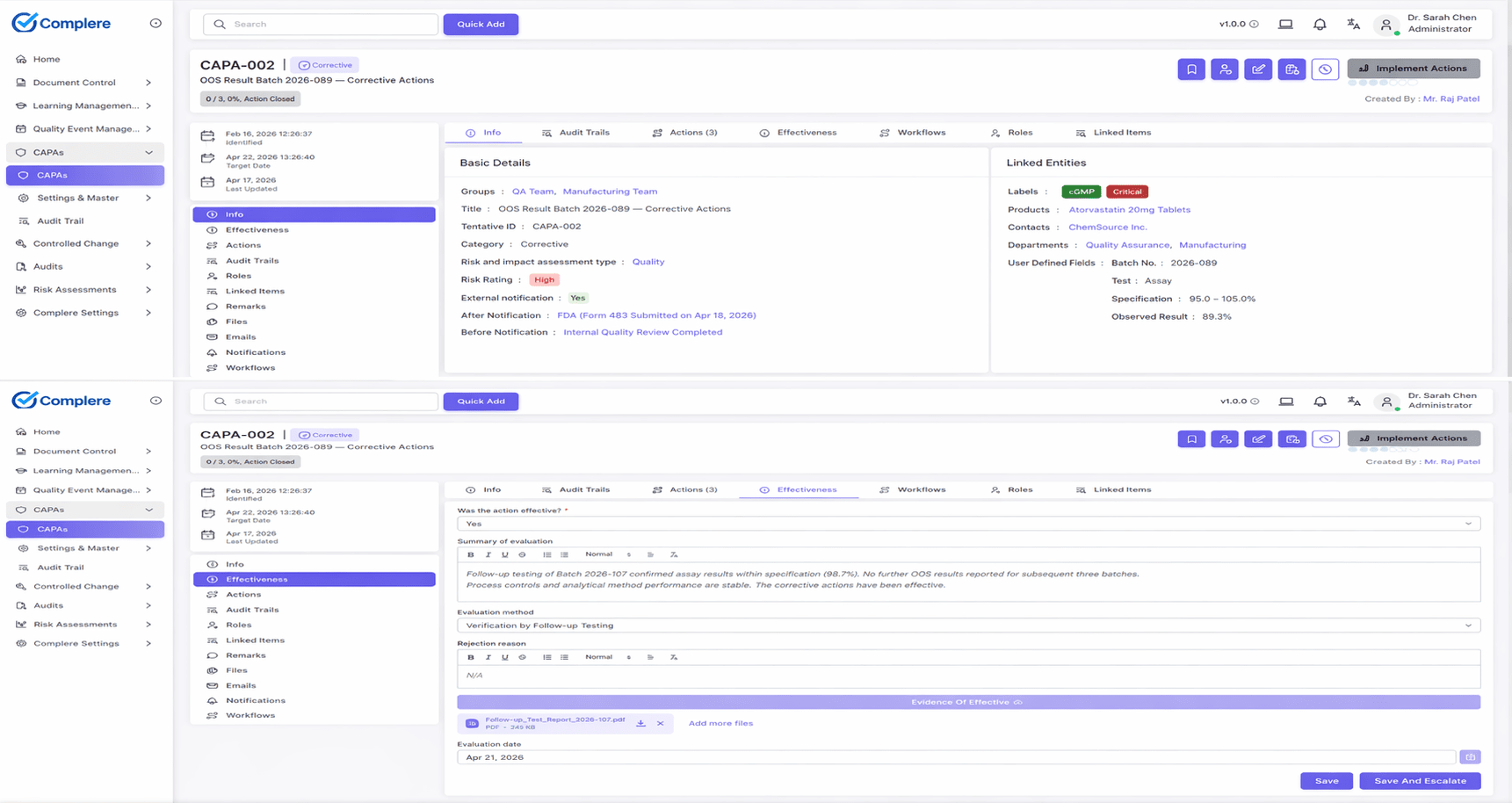
Close the loop from issue intake through investigation, action, and effectiveness verification.
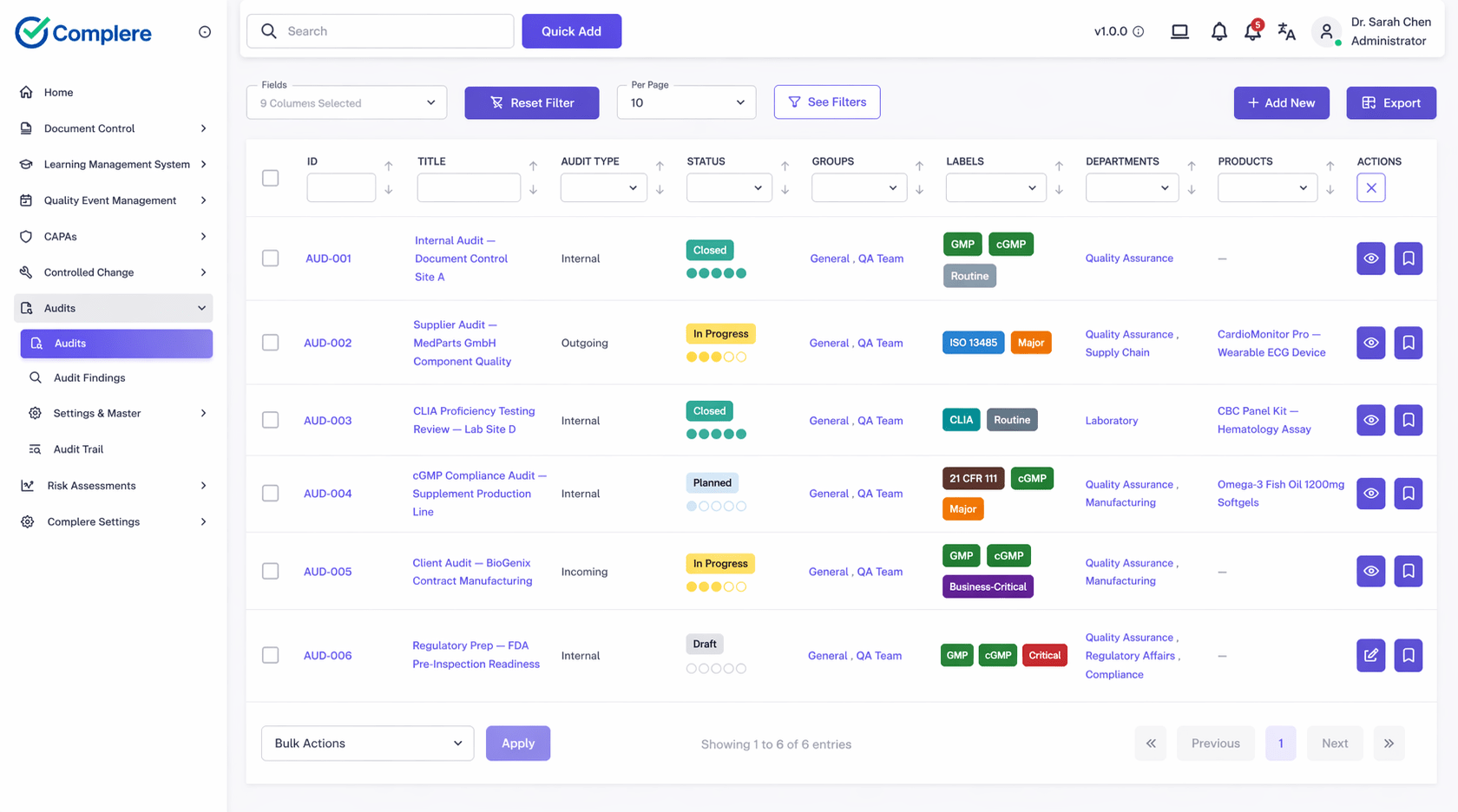
Plan internal, supplier, and site audits with evidence, findings, and follow-up in one flow.
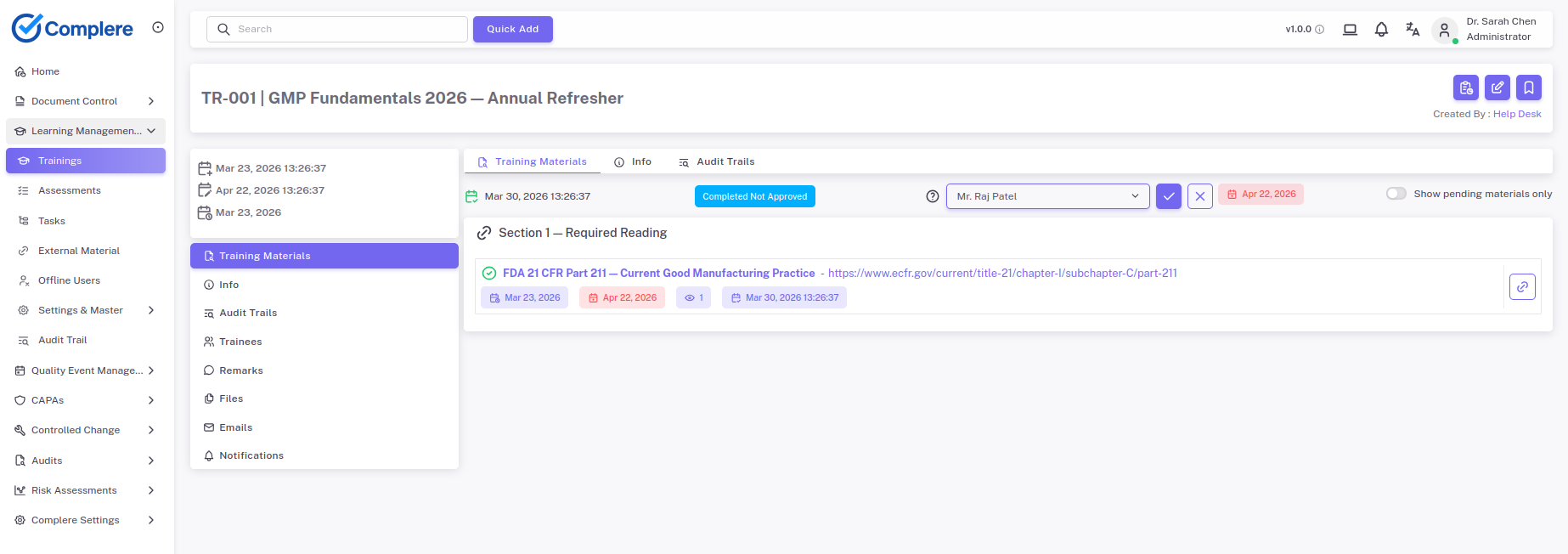
Show that people are trained, current, and assigned work that matches their role.
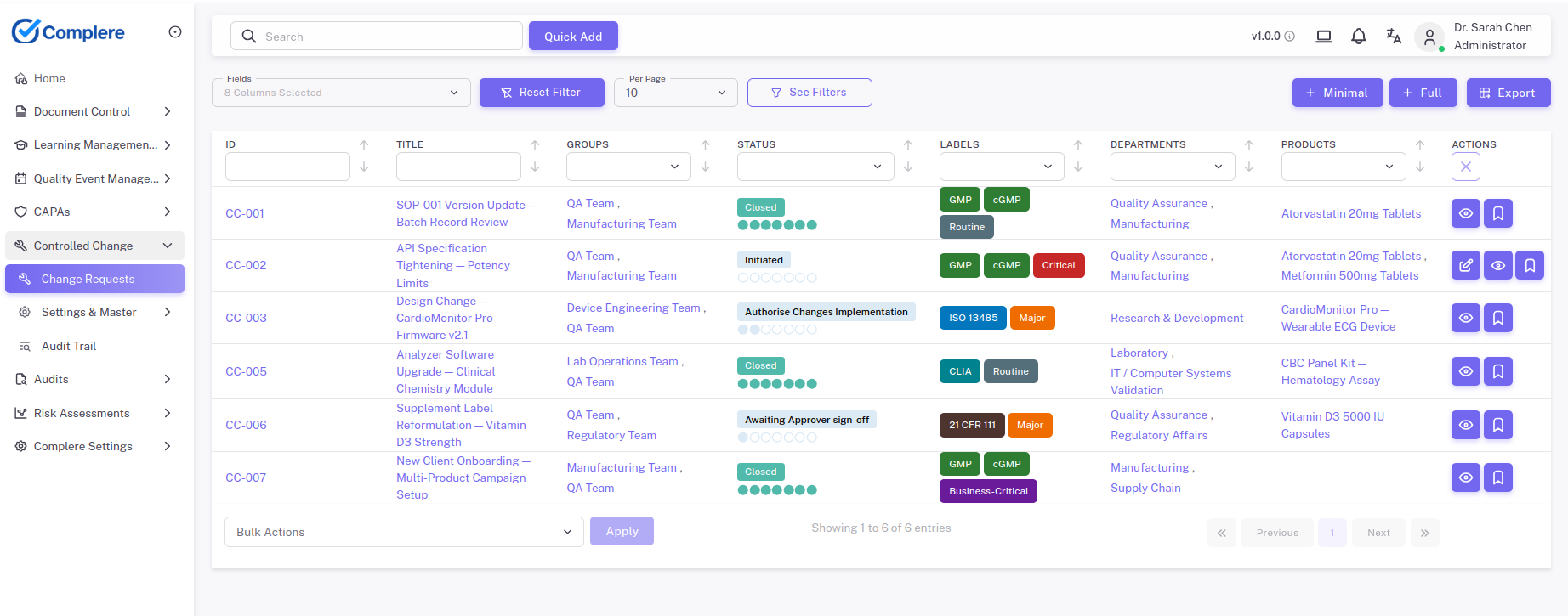
Move proposed changes through impact assessment, approval, implementation, and verification.
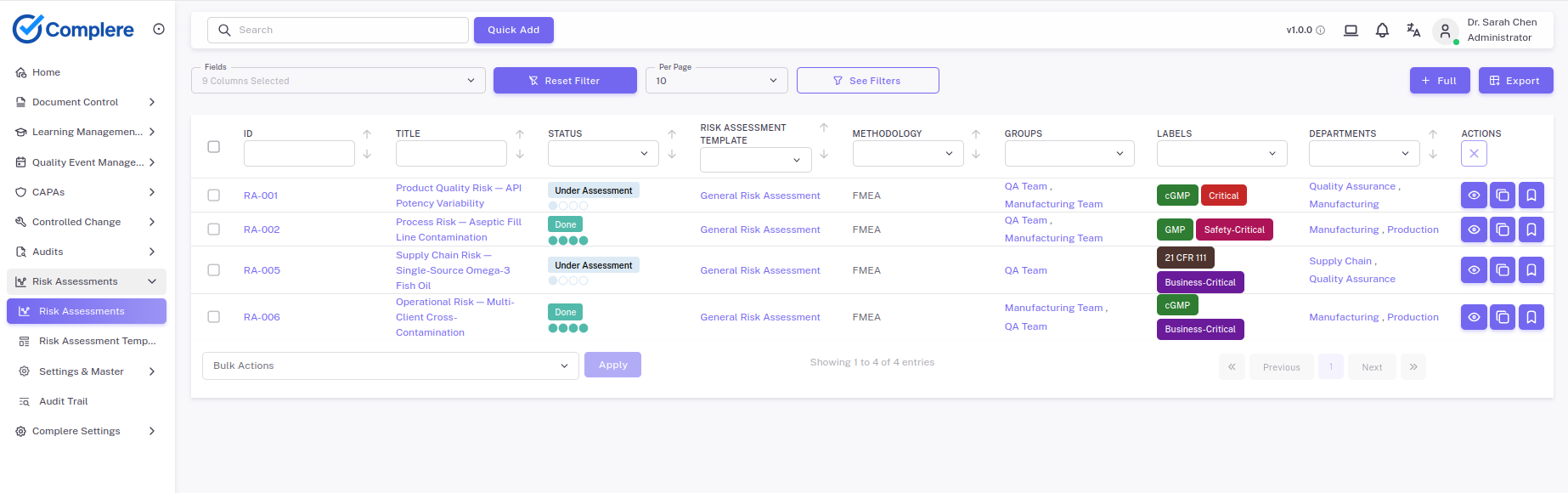
Structure risk assessments across products, processes, and quality events with scored, traceable records.
Every module includes built-in controls that support continuous compliance and defensible inspection readiness.
A staged, low-disruption path from your current state to a fully governed, inspection-ready quality system.
Requirements assessment, process mapping, and first-scope planning.
2-4 weeksWorkflow setup, role configuration, and controlled deployment prep.
4-6 weeksUser enablement, qualification activities, and acceptance checks.
3-4 weeksProduction rollout, hypercare support, and staged expansion.
OngoingWhether you are leaving paper processes, retiring spreadsheets, or replacing a legacy eQMS — Complere provides a clear path forward.

Challenge: Signatures in binders, no version history, slow retrieval in audits.
Solution & outcome: Convert controlled documents, approvals, and training records to a governed digital workflow with a full audit trail.
Explore path
Challenge: Spreadsheets break under inspection—no controls, no trail, easy to overwrite.
Solution & outcome: Replace shared-drive spreadsheets with controlled CAPA, deviation, and change workflows—inspection-ready by design.
Explore path
Challenge: Expensive, hard-to-adopt platforms that teams work around instead of in.
Solution & outcome: Migrate with a phased cutover that preserves validation posture and inspection evidence.
Explore path
Challenge: Each site runs quality its own way, so evidence never lines up.
Solution & outcome: Unified document control, CAPA, and audit across sites—with local flexibility where regulations demand it.
Explore pathRegulated organizations across pharmaceutical, medical device, and life sciences use Complere to stay inspection-ready and operationally efficient.
Structured intake, clear ownership, and escalation controls reduce time spent chasing open actions across email threads.
Parallel review routing and electronic signatures replace sequential bottlenecks and shared-drive confusion.
Automated assignments linked to document updates keep training records current and inspection-ready without manual tracking.
Whether you work in pharmaceutical manufacturing, medical devices, nutraceuticals, or contract research, Complere is configured for the specific compliance requirements of your sector.
Support GMP-centric quality operations across manufacturing, QA, validation, and release functions.
Explore pharmaceutical manufacturingConnect design, risk, CAPA, training, and document control for device-focused quality systems.
Explore medical devicesBring quality documentation, training, investigations, and review cadence into one controlled operating model.
Explore healthcare laboratoriesStandardize documents, deviations, training, and supplier-facing quality work in a growing regulated environment.
Explore nutraceuticals & supplementsCoordinate quality work across internal teams, customers, and partner sites with stronger visibility and accountability.
Explore CMO & CDMOBring controlled processes, training evidence, and follow-up visibility into distributed research operations.
Explore clinical research & CROsExplore related modules, compliance topics, and guides to build a complete picture of your quality system.
Schedule a guided demo focused on your workflows and compliance requirements, or start with a focused 30-day pilot.