Choose one workflow to improve first
Document control, CAPA, audit follow-up, or another high-value process.
A Complere pilot is a structured 30-day engagement built around one high-value workflow — document control, CAPA, audits, or another priority. You define success, we configure the environment, and together we prove fit before any broader commitment. Records in the pilot carry the same ALCOA+ data integrity expectations — attributable, time-stamped history — as production use; see data integrity and audit trails for the platform posture.
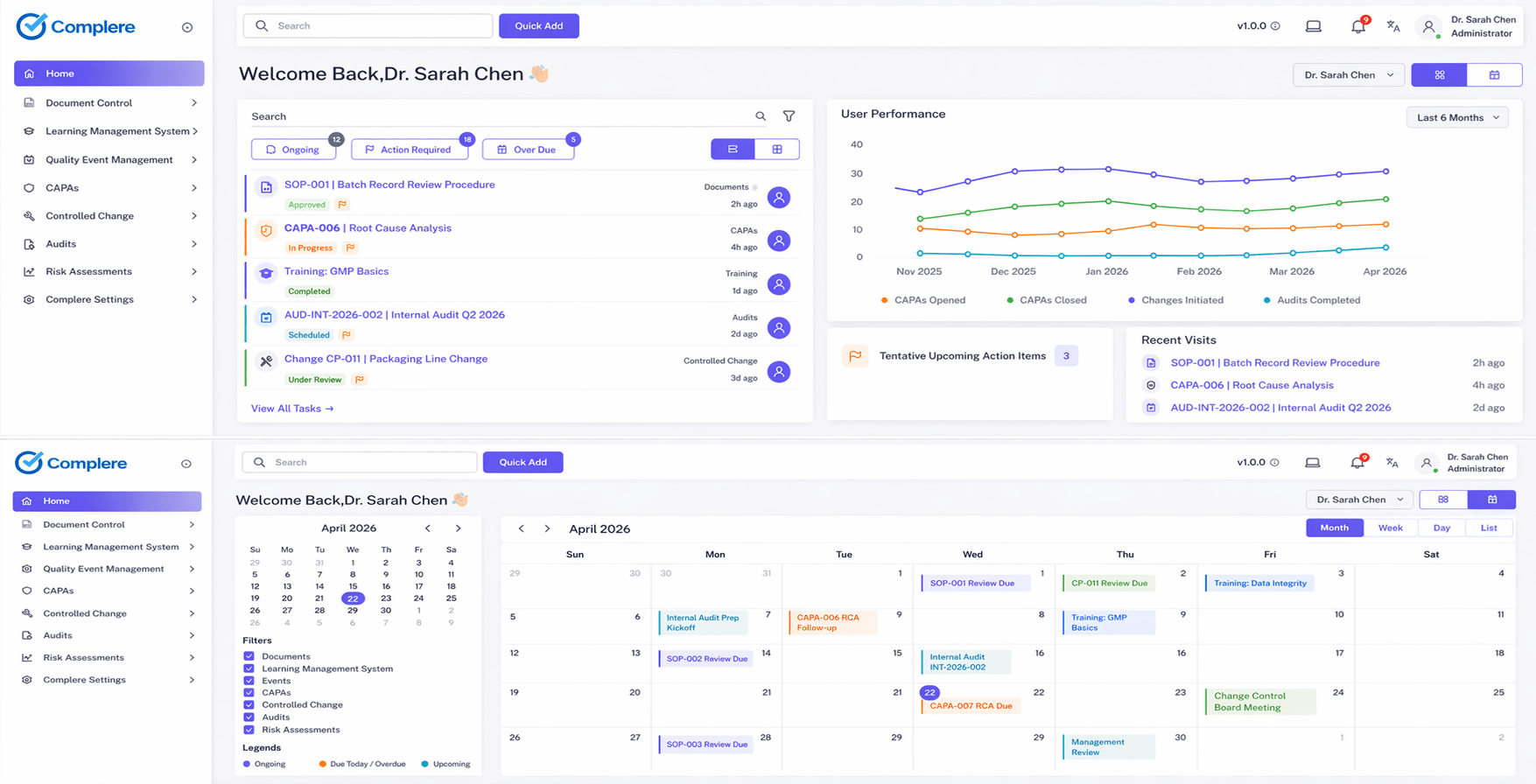
A focused 30-day pilot works best when the first workflow is well defined, success can be measured, and the right people are available to review and approve during the engagement.
Built for regulated life-sciences teams that need control, auditability, and practical adoption.
Document control, CAPA, audit follow-up, or another high-value process.
Cycle time, visibility, retrieval, and adoption metrics make the first phase concrete.
A good pilot page should naturally tee up broader module or site rollout.
Tell us about your quality priorities and we will get back to you within one business day.
Request a 45-minute demo and we will show you the exact workflows and compliance controls relevant to your team.