Compliance built into every workflow
Validation, data integrity, audit trail, and access controls are implemented at the platform level — not configured separately per module or added as afterthoughts.
Complere maps FDA 21 CFR Part 11, EU GMP Annex 11, ISO 13485, ALCOA+, and GAMP 5 to the controls and evidence your teams actually produce: signatures, audit trails, training completion, change history, and retrieval under inspection. Regulatory language shows up where decisions happen, so QA and operations share one narrative.
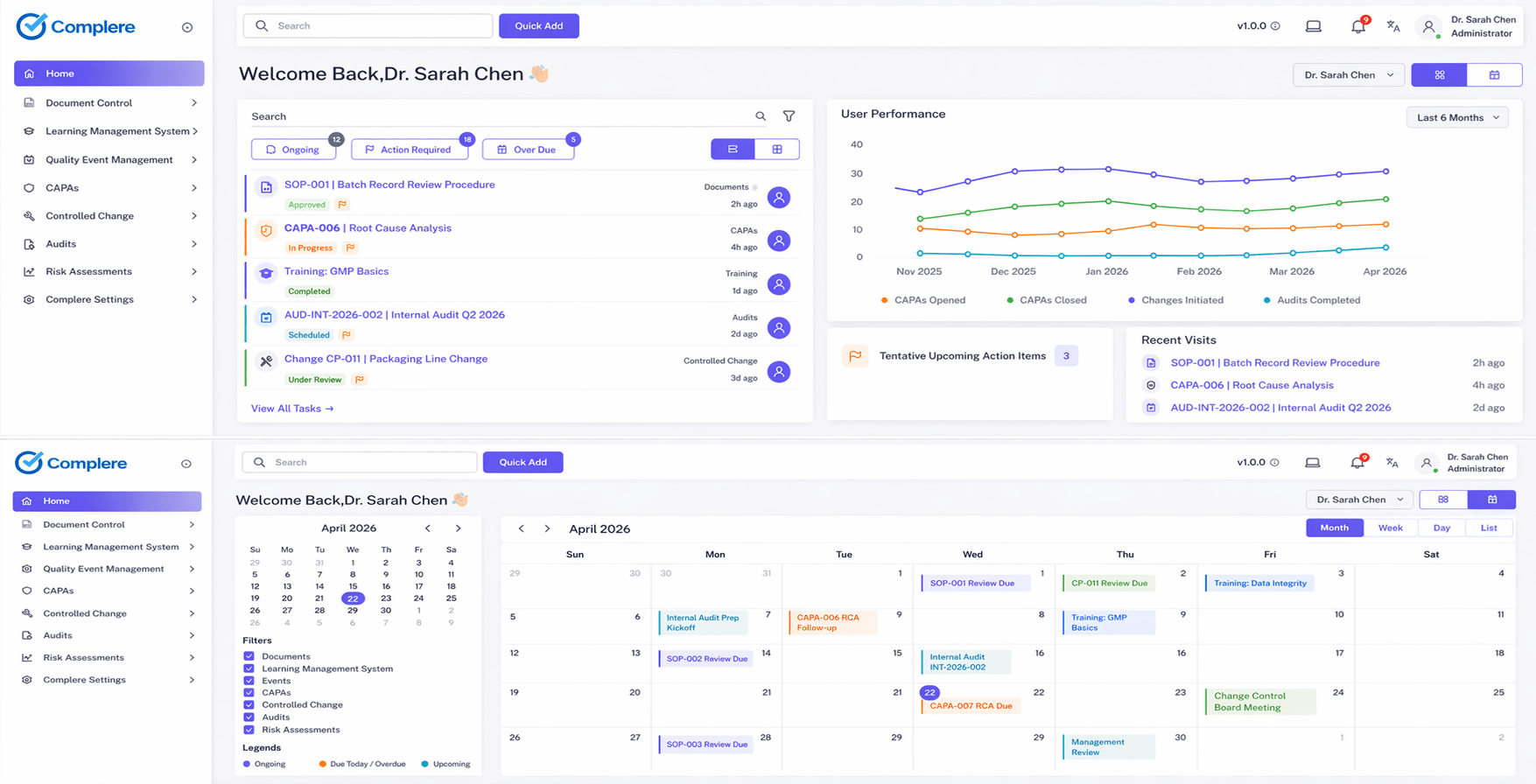
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Validation, data integrity, audit trail, and access controls are implemented at the platform level — not configured separately per module or added as afterthoughts.
Part 11, Annex 11, ALCOA+, and validation lifecycle concepts are explained in terms quality teams and commercial buyers can evaluate and communicate.
Complere provides the posture documentation, technical control overview, and compliance resources that support due diligence and qualification conversations.
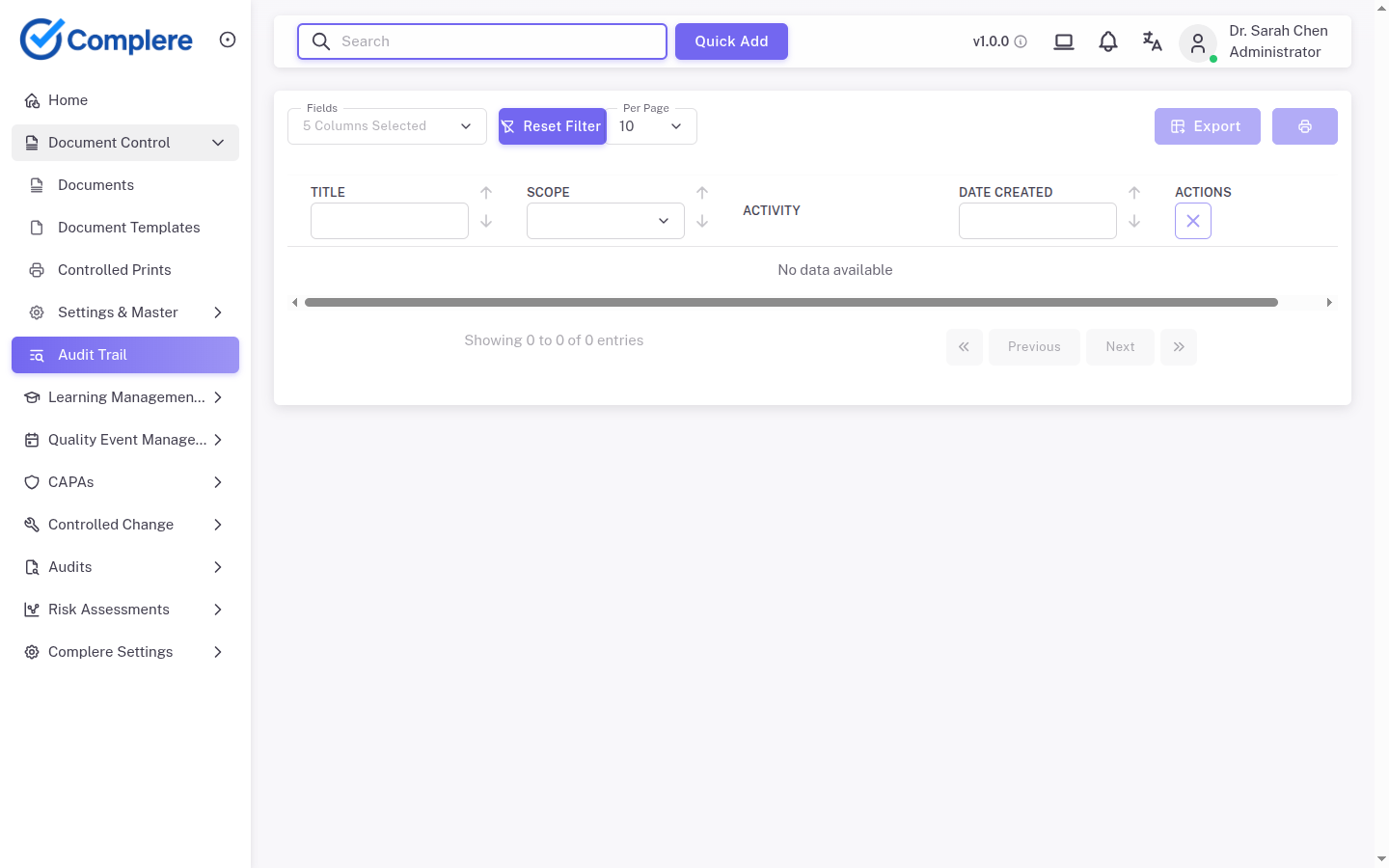
Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
A document is revised, a CAPA is raised, an audit finding is logged — the workflow is governed from the point of capture.
Attribution, timestamping, approval routing, and access scoping happen as part of the workflow — not as a separate compliance step.
The audit trail, signed record, and related workflow history stay accessible and linked — no after-the-fact reconstruction needed.
Quality teams can retrieve, export, and present compliance evidence to inspectors in minutes, with full context.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Every key requirement — Part 11 §11.10, Annex 11 clauses, ALCOA+ principles — maps to the specific platform controls that address it.
Audit trail, electronic signatures, access control, and change governance work the same way across all modules.
Complere provides a validation-ready implementation package including VMP templates, URS frameworks, and IQ/OQ/PQ test scripts.
The compliance hub connects platform proof to long-form regulatory explainers, glossary definitions, and downloadable templates.
Our compliance team walks through Part 11, Annex 11, data integrity, and validation controls during a tailored 45-minute demo.