Teams and partners operate across locations
Consistency is hard when training, documentation, and investigations are spread across groups.
CRO and clinical research teams need GCP-aligned quality controls: current procedures, role-based training evidence, deviations and CAPA discipline, and sponsor or regulator audit readiness across distributed teams and trial sites. Qualification and read-and-understand records for monitors and investigators stay current-state controlled — see training and competency.

The quality and compliance pressures unique to your regulated market — and how Complere is built to address them.
Consistency is hard when training, documentation, and investigations are spread across groups.
Evidence behind approvals, follow-up actions, and training records needs to be accessible in minutes — not hours spent reconstructing from multiple systems.
Process updates are only useful if teams can prove the new expectations were rolled out.
The regulatory frameworks, guidance documents, and audit expectations that govern quality operations in this sector.
2024 revised GCP • Risk-based quality management • Enhanced sponsor oversight requirements • Quality tolerance limits for trial conduct
Investigational new drug applications • Safety reporting • Site monitoring documentation
EU Clinical Trials Regulation • Mandatory for EU trial authorisation since January 2023 • Harmonised submission and safety reporting across member states
The Complere modules most commonly used by teams in this industry to manage compliance and quality operations.
Assign and track GCP training by protocol, role, and site — with electronic sign-off and competency records that satisfy ICH E6 R3 investigator and staff qualification requirements under FDA 21 CFR 312.
Learn moreMaintain controlled protocol documents, SOPs, and essential study documents with Part 11–compliant approval workflows and version-controlled effective dates — keeping trial master files inspection-ready.
Learn moreSchedule internal quality audits, vendor audits, and inspection readiness reviews with structured checklists, finding classification, and CAPA linkage aligned to ICH GCP audit program expectations.
Learn moreRoute protocol amendments, SOP revisions, and system changes through impact-assessed approval workflows — with linked training triggers and audit trail documenting who approved what and when.
Learn moreExplore related modules, compliance topics, and guides to build a complete picture of your quality system.

Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
Explore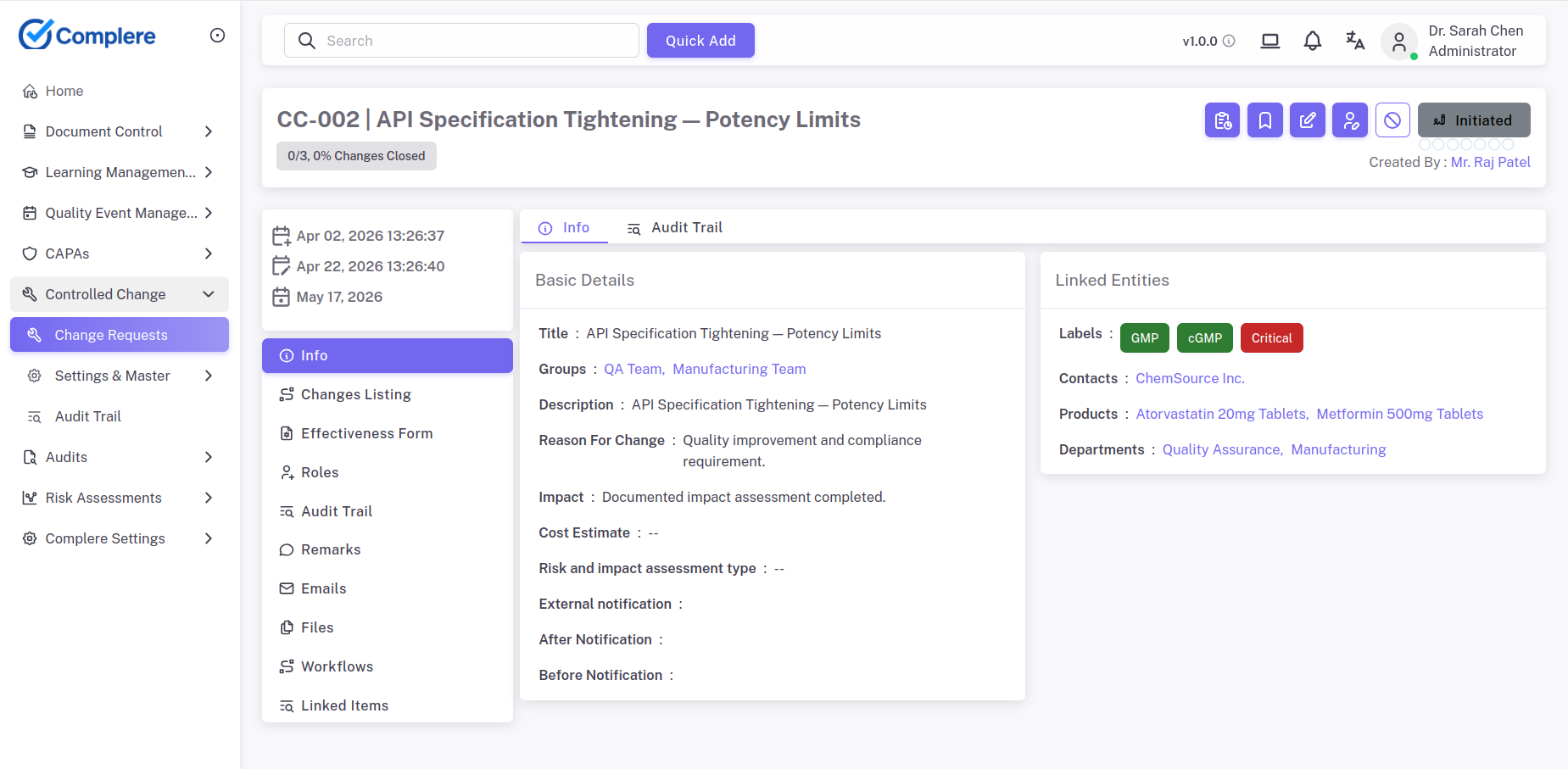
Explore this topic in more depth to build a complete picture of your quality and compliance operations.
ExploreOur demos cover training rollout, procedure management, deviation handling, and audit follow-up for distributed research operations.