Keep the audit program visible
Schedules, scopes, checklists, and assigned auditors stay centralized.
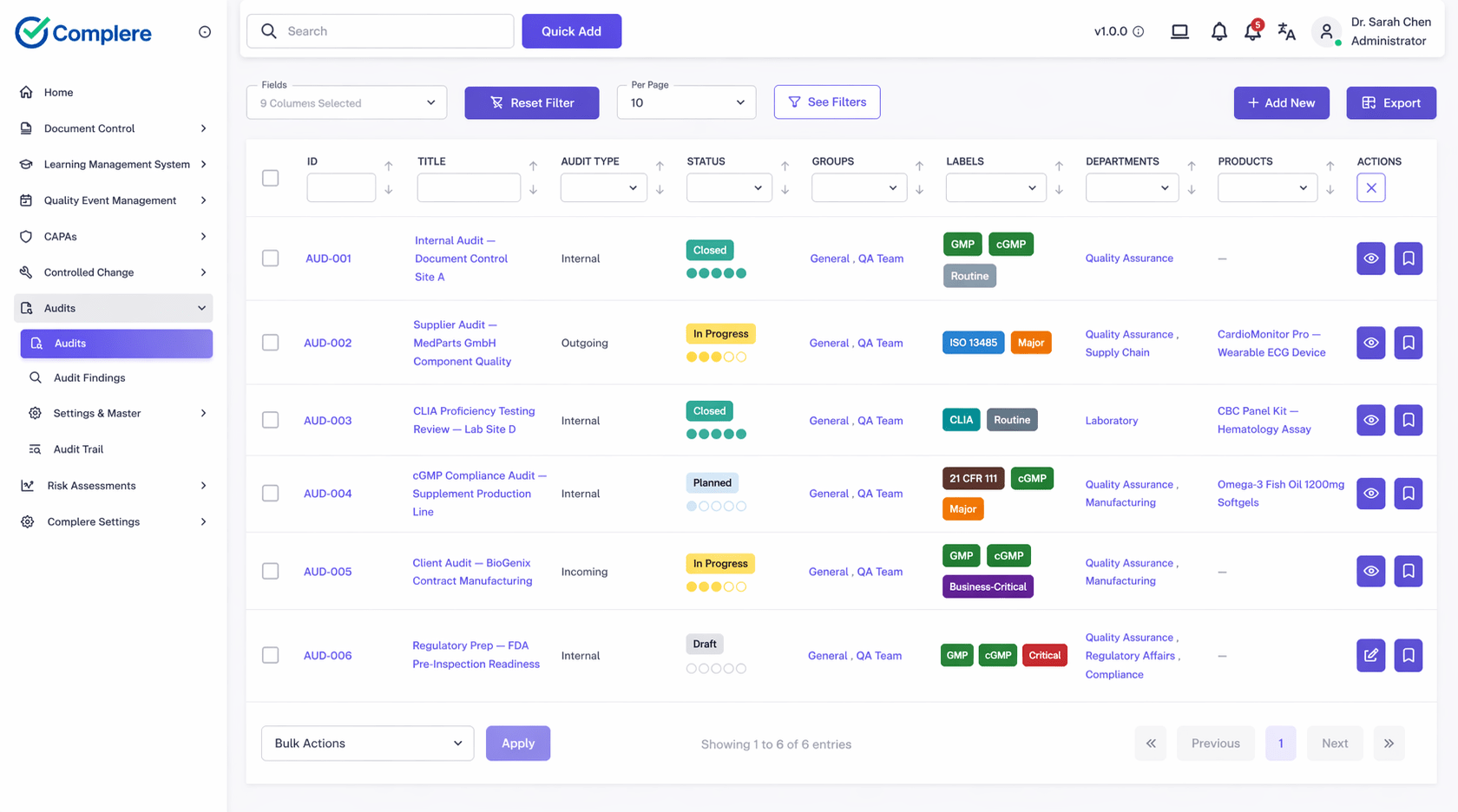
Plan internal, supplier, and regulatory-style audits with schedules and checklists; record findings with severity and ownership; route corrective actions; and close only when evidence attachments and verification steps are complete — so the full story from planning through closure is in one system.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Schedules, scopes, checklists, and assigned auditors stay centralized.
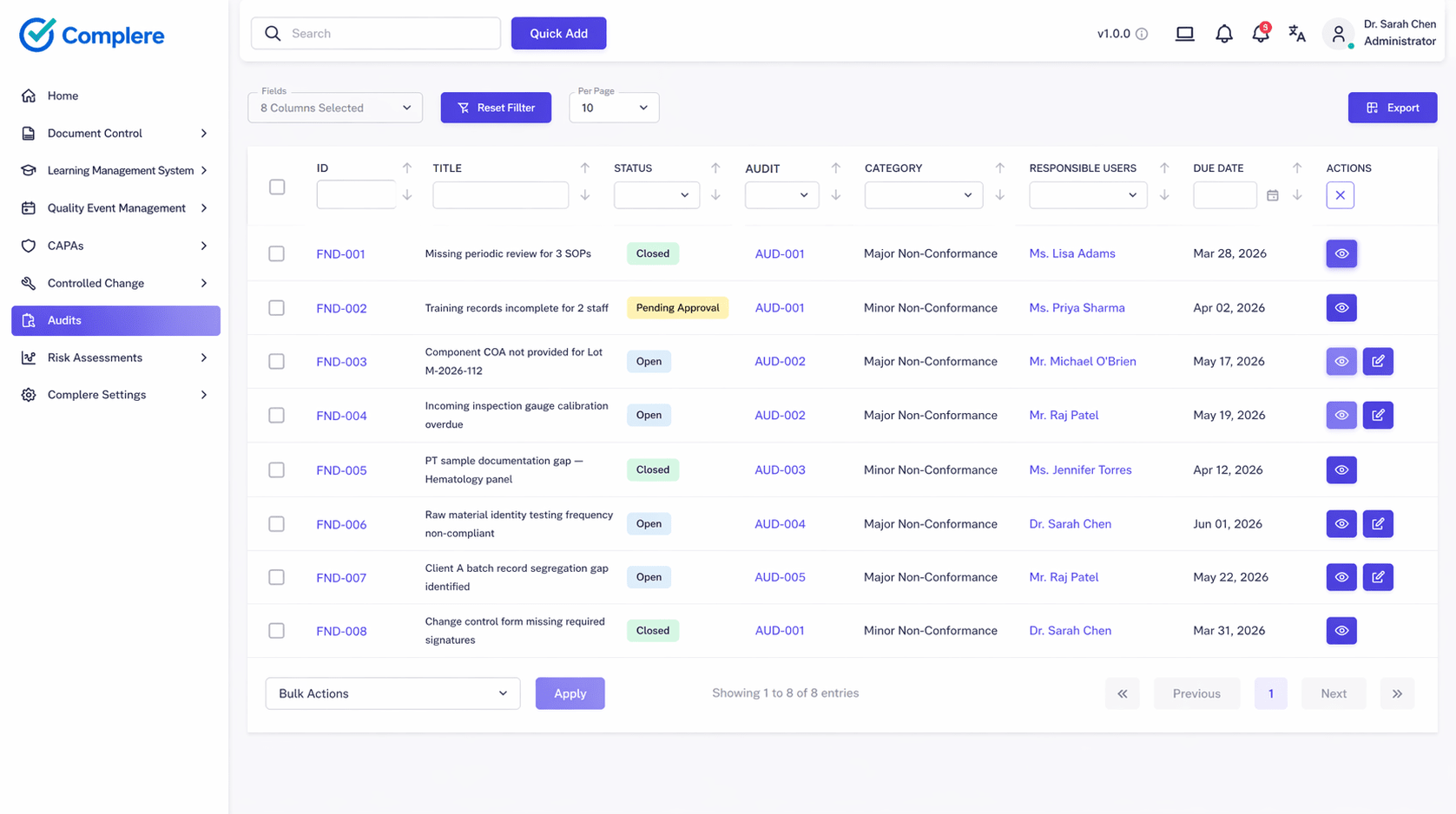
Tie observations to categories, severity, owners, and related quality records.
Connect corrective actions and verification steps instead of relying on email threads.
Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
Define scope, auditees, checklists, and scheduling windows.
Capture evidence and findings while the audit is underway.
Route remediation or CAPA ownership to the right functions.
Confirm evidence before closing findings and reporting outcomes.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Keep recurring internal and supplier programs structured and repeatable.
Classify issues and route them directly into remediation workflows.
Keep follow-up visible from finding to closure evidence.
Summaries and dashboards show open findings, aging, and recurring themes.
When audit programs cover IT systems, teams often pair audit evidence with the platform’s validation approach — IQ/OQ/PQ or equivalent, change control for releases, and testing support — so findings about the quality system and the system of record stay aligned.
The audit management page is designed to connect directly into CAPA, dashboards, and industry proof pages.