Explain role qualification clearly
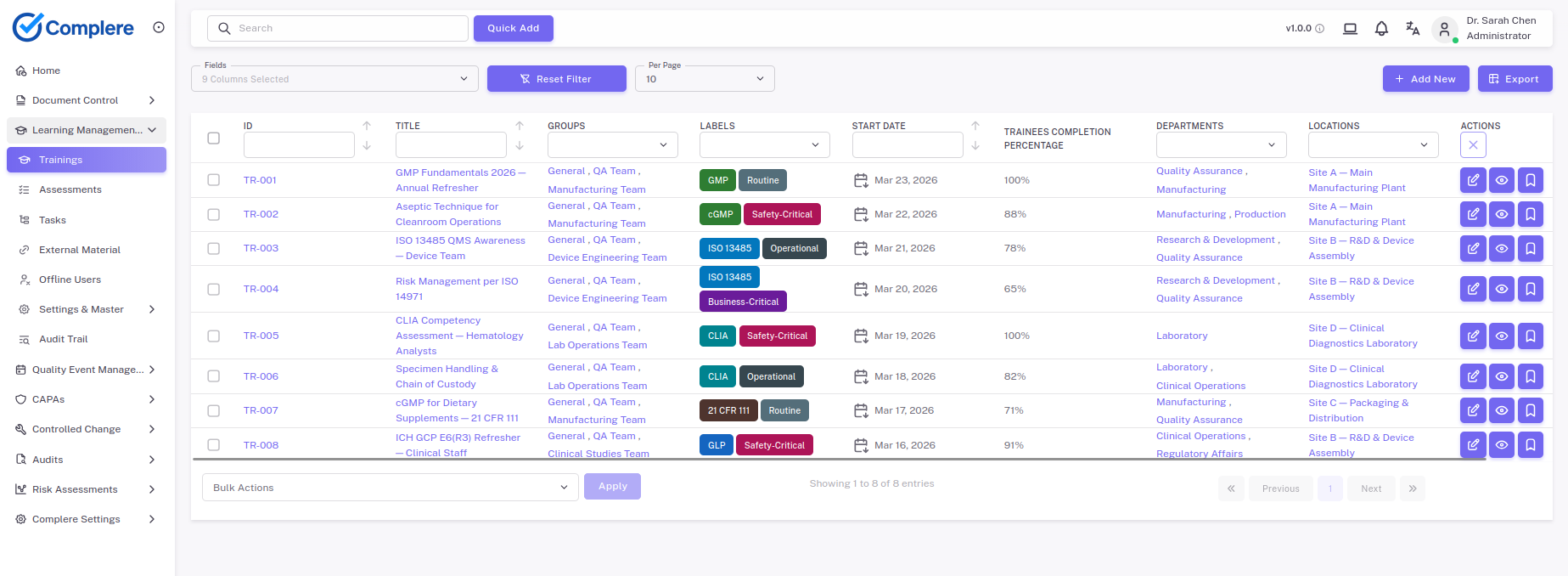
Tie curricula and task requirements to departments, sites, and job functions.
Bring curricula, assessments, qualification evidence, retraining triggers, and training history into one competency narrative that supports GMP, device, and laboratory environments.
How this module improves quality operations, reduces compliance risk, and supports inspection readiness for regulated teams.
Tie curricula and task requirements to departments, sites, and job functions.
Document updates and workflow changes can feed new assignments without manual chase-down.
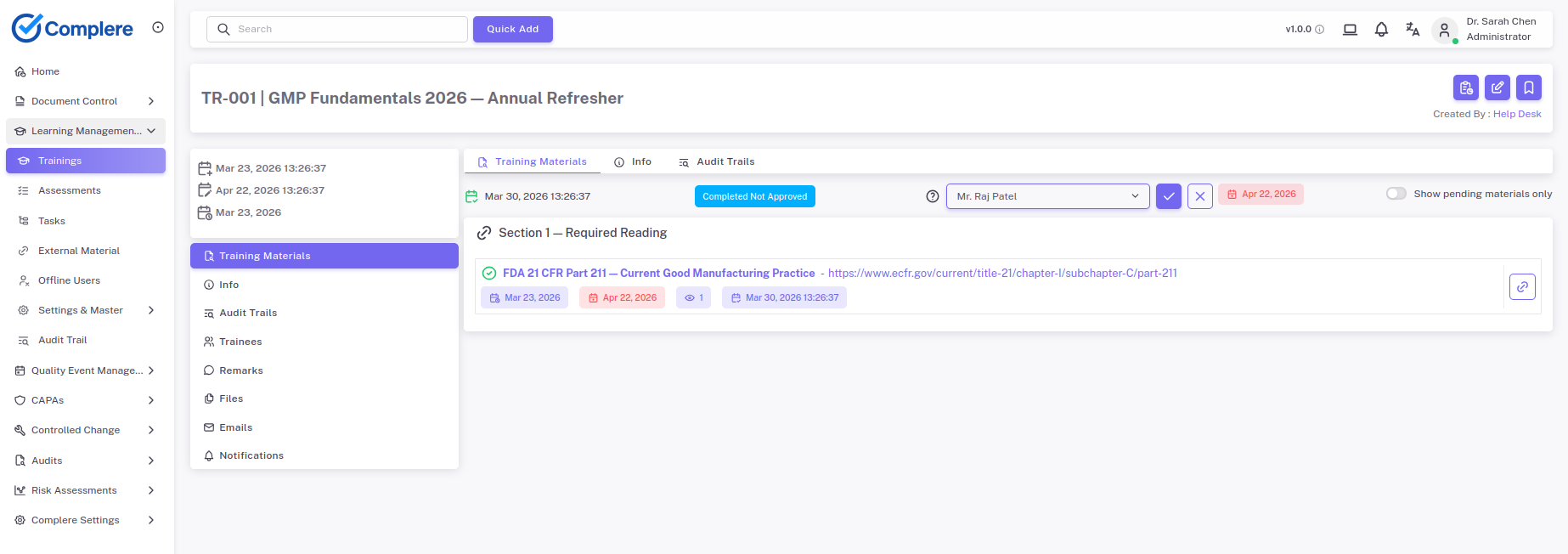
Show current and historical completion data, assessments, and gaps from one place.
Each step in this workflow is supported by governed records, electronic signatures, and a full audit trail — so quality operations stay traceable and inspection-ready.
Map curricula and qualifications to teams, jobs, and procedures.
Publish required learning with due dates and reminders.
Capture completion, assessment results, and supervisor acknowledgement.
Use dashboards to monitor overdue or incomplete requirements.
The specific controls, features, and workflow functions built into this module to support your quality and compliance objectives.
Organize required, optional, and recurring learning by role and function.
Support assessments, acknowledgements, and externally delivered training records.
Expose overdue assignments, qualification risk, and completion rates.
Keep the connection between approved content and human competency visible.
Training pages link naturally into document control, change control, and management review proof points.