What buyers are usually trying to confirm
Most teams are not looking for abstract compliance claims. They want to know whether the system produces controlled records, maintains a readable history, limits access appropriately, and ties sign-off events to specific actions.
- Can the system show who approved what and when?
- Can we retrieve record history quickly during inspection?
- How are signatures and user roles handled in daily workflows?
How Complere supports Part 11 compliance
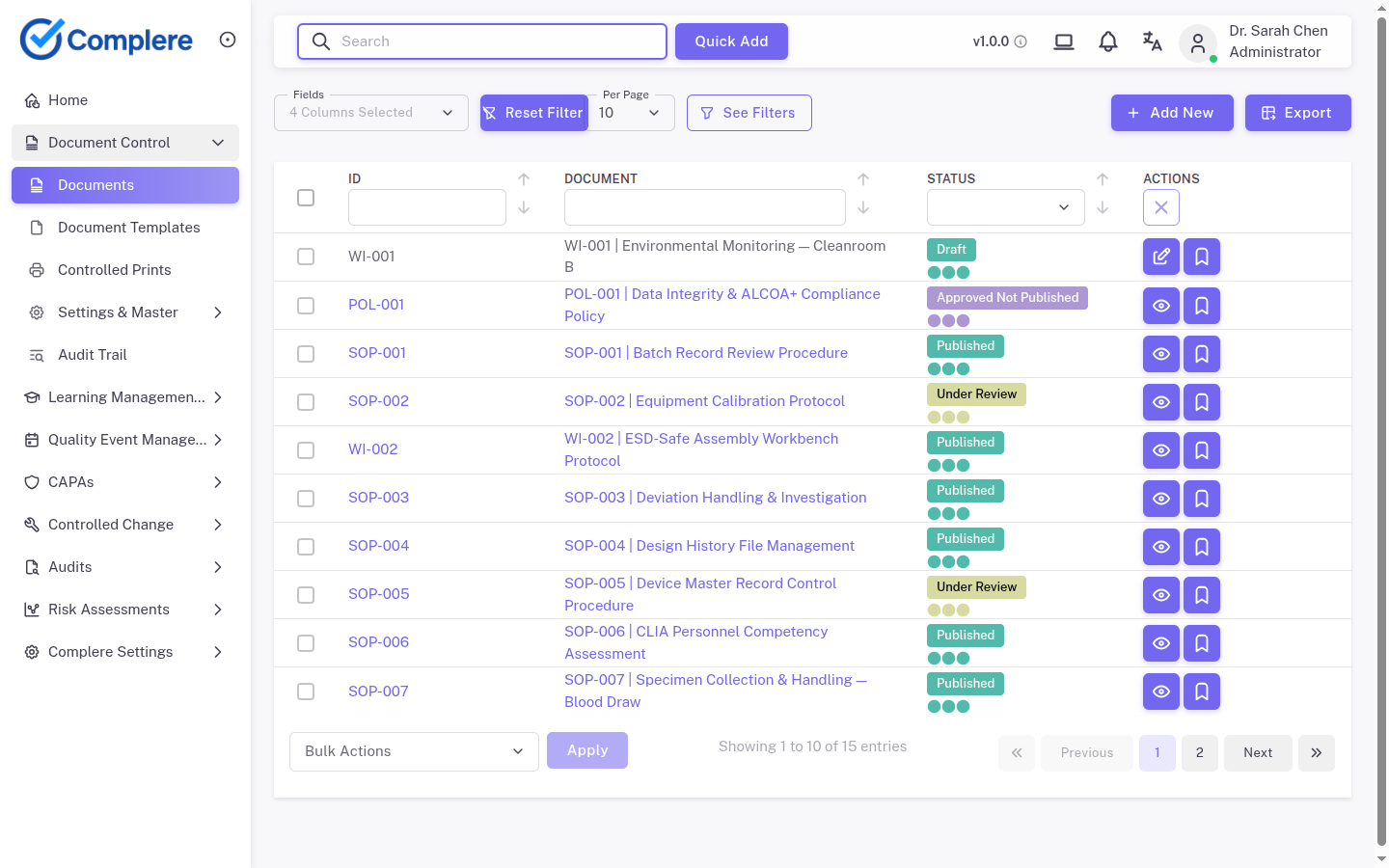
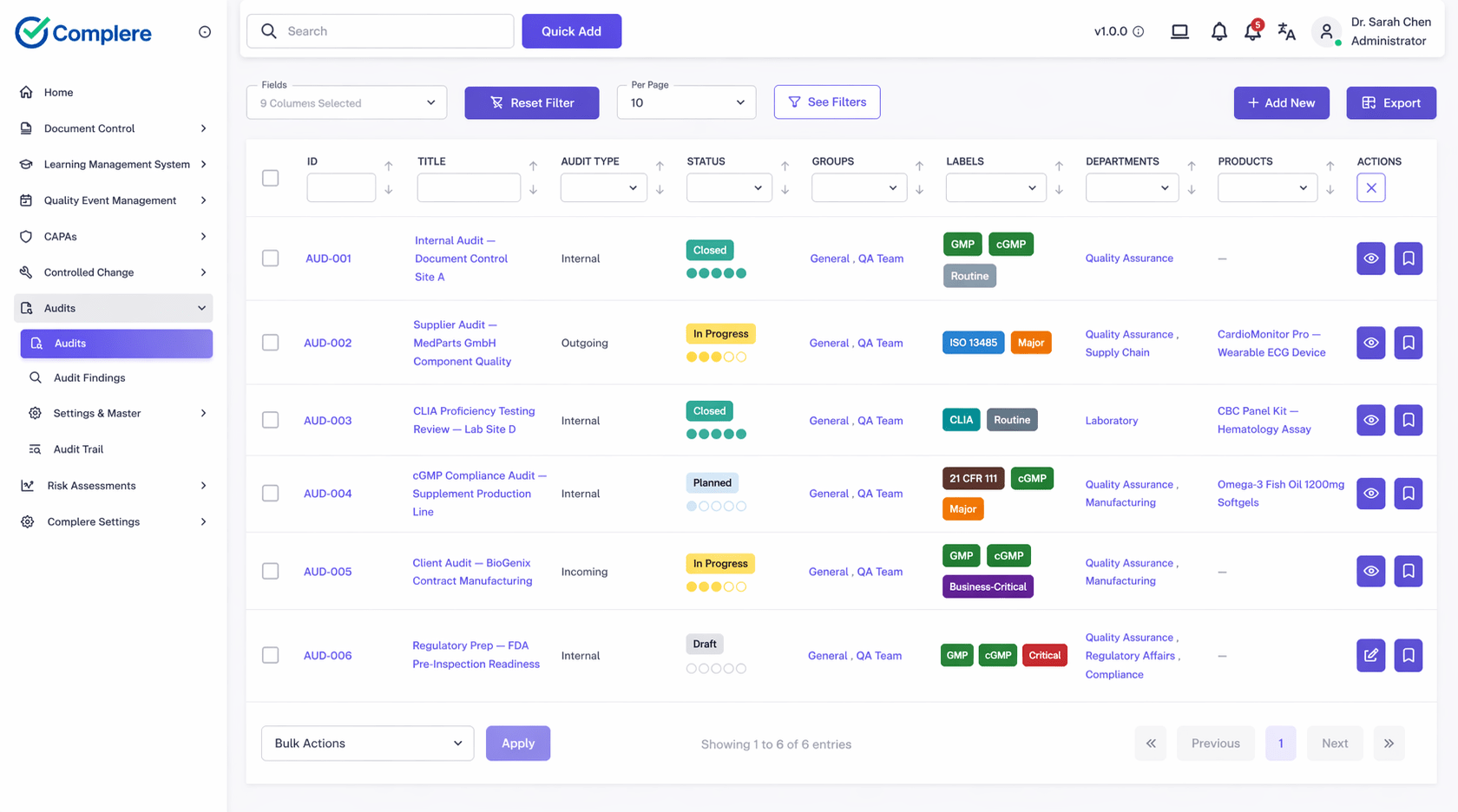
Complere connects Part 11 requirements to daily quality operations: role-based access controls limit who can create, review, and approve records. Workflow sign-off points capture electronic signatures with re-authentication and intent. Time-stamped audit trails record every action, change, and access event across documents, CAPA, audits, training, and change control.
Electronic records, electronic signatures, audit trails, access controls, and system validation — Complere addresses all five core Part 11 control areas in a single governed platform.
Related compliance resources
For teams doing deeper Part 11 due diligence, Complere's compliance hub provides detailed mapping tables, signature workflow explanations, and data integrity controls aligned to ALCOA+ principles.
- Electronic Records & Signatures — detailed control mapping
- Data Integrity & Audit Trails — ALCOA+ posture
- Validation Approach — CSV package for Part 11 systems